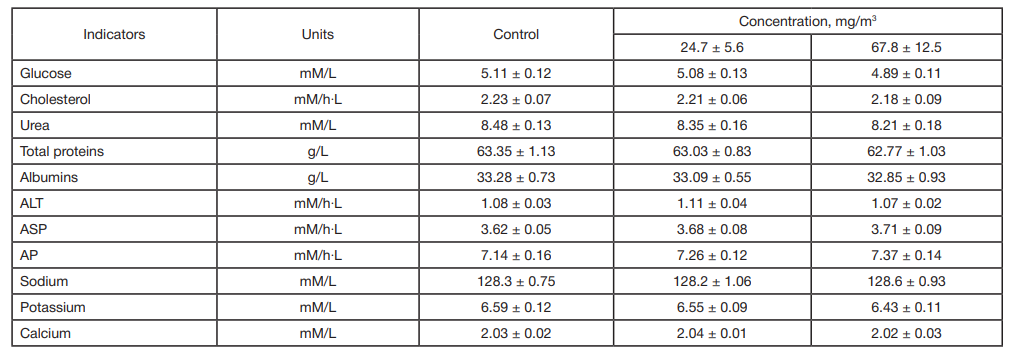
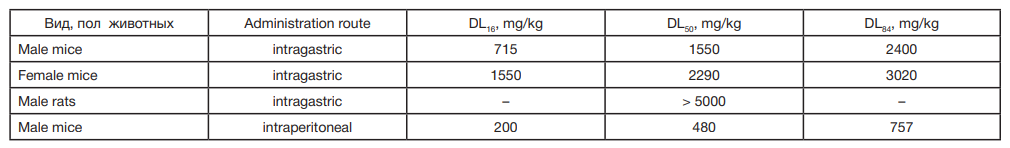
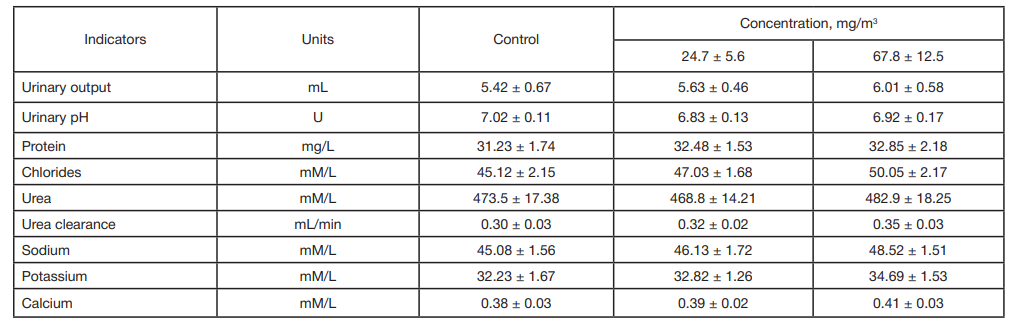
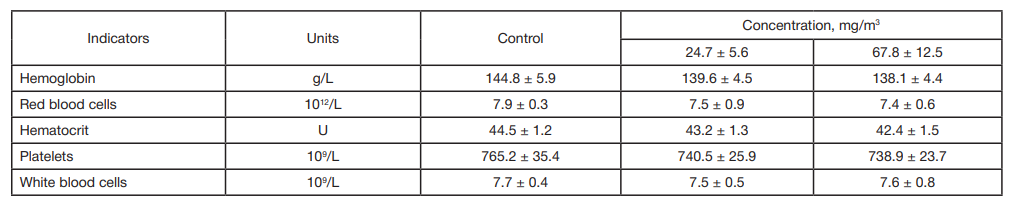
This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (CC BY).
ORIGINAL RESEARCH
Assessing toxicity and hazard of agidol-1, the vitamin E synthetic analogue
1 All-Russian Research Center for the Safety of Bioactive Substances, Staraya Kupavna, Moscow region, Russia
2 Pirogov Russian National Research Medical University, Moscow, Russia
3 Institute of Disinfectology, Erisman Federal Scientific Centre of Hygiene of Rospotrebnadzor, Moscow, Russia
Correspondence should be addressed: Natalia I. Sheina
Ostrovityanov, 1, Moscow, 117513, Russia; ur.liam@aniehs_in
Author contribution: Golubeva MI — describing the results, literature review, manuscript writing; Sheina NI — literature review, describing the results, manuscript writing and formatting; Bidevkina MV — describing the results, manuscript w, processing and describing the study results; Bobrineva IA — testing, processing and describing the study results; Fedorova EA — experimental procedure, processing the study results.
Compliance with ethical standards: the experimental study was compliant with the necessary legal acts (Declaration of Helsinki 2013, GOST 33044-2014 “Principles of Good Laboratory Practice”; Order of the Ministry of Health of the RF No. 188n of 01.04.2016 “Rules of Good Laboratory Practice”). The study was approved by the Ethics Committee of the Pirogov Russian National Research Medical University (protocol No. 10/23 dated 15 May 2023). Correspondence should be addressed: Natalia I. Sheina